Bring Vaccines to Market Faster
Validated and standardized kits enable QC departments to quickly achieve global quality initiative
World Vaccine Congress | March 30-April 2, 2026 | Washington, D.C.Find us at the SSI Diagnostica Group Booth
Streamlining process development of RNA capping using VaxArray 5’CapQ and HPLC-based methods.

Rebecca Young, PhD
Sr Scientist II, RNA Sciences
Aldevron
April 1, 12.55 pm ET
For mRNA drug substance to be safe and efficacious it must be highly capped. Yet measuring the capping efficiency of RNA is a technical challenge. Here we present several case studies of using InDevR’s VaxArray instrument as a high-throughput capping efficiency and integrity assay in partnership with HPLC-based capping efficiency methods. This combination allows streamlining of workflows to facilitate process development around enzymatic and co-transcriptional capping. Projects can then easily move from R&D to research-grade manufacturing, with clear paths into GMP manufacturing.
To learn more about using the VaxArray to measure capping efficiency and mRNA integrity, download this GEN tutorial authored by Erica Dawson, PhD and Rebecca Young, PhD.
Maximize Efficiency in Vaccine Development with VaxArray
Discover the VaxArray platform, your solution in multiplexed immunoassays designed to streamline vaccine development and analysis. This state-of-the-art system offers unparalleled precision and efficiency, empowering researchers to achieve faster, more reliable results in their efforts to advance public health.
- High-Efficiency Testing – improve at-line analysis with multiplexed immunoassays.
- Simplify Standardization – 21 CFR Part 11 compatible software and easy-to-use kits simplify standardization across workflows.
- Versatile Platform – analyze a wide range of vaccine components, from antibodies and antigens to nucleic acids.
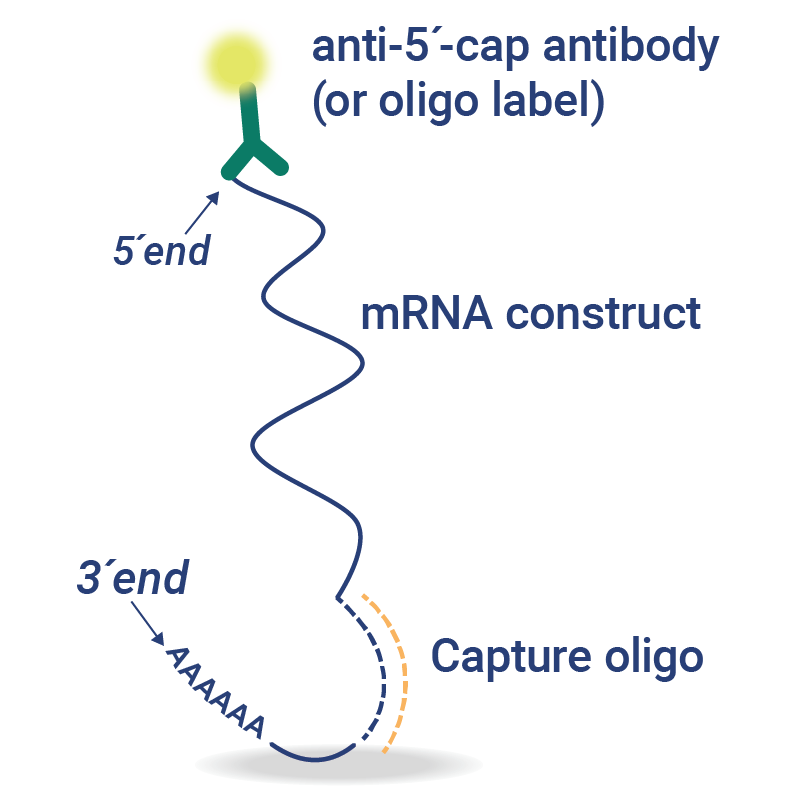
5’CapQ Assay: Simplify Measurement of mRNA Attributes
The 5′ cap and poly(A) tail elements of mRNA are essential for protein expression and mRNA stability. Waiting for the mRNA analysis of concentration, intactness, and purity from a central laboratory or outsourced partner slows down mRNA vaccine development.
In just 90 minutes, the 5’CapQ Assay is a simple, direct measurement of capped and tailed mRNA. This assay works by capturing the mRNA molecules via the 5’ cap and labeling the molecules on the poly(A) tail, enabling quantification of only the complete intact mRNA molecules. RNA molecules that lack a 5’ cap or poly(A) tail are not measured in the 5’CapQ Assay, leading to a single test for quantifying your mRNA.
- Develop Vaccines Faster – Consolidate your mRNA capping and intactness testing into 1 assay that can be completed in less than two hours.
- Analyze at Your Benchtop – Reduce testing bottlenecks by bringing mRNA analytics to your facility.
- Simplify Workflow Process – compatible with LNP formulations eliminating the need for laborious extraction procedures.